1.1 Early Ideas in Atomic Theory
Learning Objectives
By the end of this section, you will be able to:
- State the postulates of Dalton’s atomic theory.
- Use postulates of Dalton’s atomic theory to explain the laws of definite and multiple proportions.
The language used in chemistry is seen and heard in many disciplines, ranging from medicine to engineering to forensics to art. The language of chemistry includes its own vocabulary as well as its own form of shorthand. Chemical symbols are used to represent atoms and elements. Chemical formulas depict molecules as well as the composition of compounds. Chemical equations provide information about the quality and quantity of the changes associated with chemical reactions.
The beginning of this text will lay the foundation for our study of the language of chemistry. The concepts of this foundation include the atomic theory, the composition and mass of an atom, the variability of the composition of isotopes, ion formation, chemical bonds in ionic and covalent compounds, the types of chemical reactions, and the naming of compounds. We will also introduce one of the most powerful tools for organizing chemical knowledge: the periodic table.
Atomic Theory Through the 19th Century
The earliest recorded discussion of the basic structure of matter comes from ancient Greek philosophers, the scientists of their day. In the 5th century BC, Leucippus and Democritus argued that all matter was composed of small, finite particles that they called atomos, a term derived from the Greek word for “indivisible.” They thought of atoms as moving particles that differed in shape and size, and which could join together. Later, Aristotle and others came to the conclusion that matter consisted of various combinations of the four “elements”—fire, earth, air, and water—and could be infinitely divided. Interestingly, these philosophers thought about atoms and “elements” as philosophical concepts, but apparently never considered performing experiments to test their ideas.
The Aristotelian view of the composition of matter held sway for over 2000 years, until English schoolteacher John Dalton helped to revolutionize chemistry with his hypothesis that the behaviour of matter could be explained using an atomic theory. First published in 1807, many of Dalton’s hypotheses about the microscopic features of matter are still valid in modern atomic theory. Here are the postulates of Dalton’s atomic theory:
- Matter is composed of exceedingly small particles called atoms. An atom is the smallest unit of an element that can participate in a chemical change.
- An element consists of only one type of atom, which has a mass that is characteristic of the element and is the same for all atoms of that element (Figure 1). A macroscopic sample of an element contains an incredibly large number of atoms, all of which have identical chemical properties.

Figure 1. A pre-1982 copper penny (left) contains approximately 3 × 1022 copper atoms (several dozen are represented as brown spheres at the right), each of which has the same chemical properties. (Credit: modification of work by “slgckgc”/Flickr) CC BY 4.0 - Atoms of one element differ in properties from atoms of all other elements.
- A compound consists of atoms of two or more elements combined in a small, whole-number ratio. In a given compound, the numbers of atoms of each of its elements are always present in the same ratio (Figure 2).

Figure 2. Copper(II) oxide, a powdery, black compound, results from the combination of two types of atoms—copper (brown spheres) and oxygen (red spheres)—in a 1:1 ratio. (Credit: modification of work by “Chemicalinterest”/Wikimedia Commons) CC BY 4.0 - Atoms are neither created nor destroyed during a chemical change, but are instead rearranged to yield substances that are different from those present before the change (Figure 3).
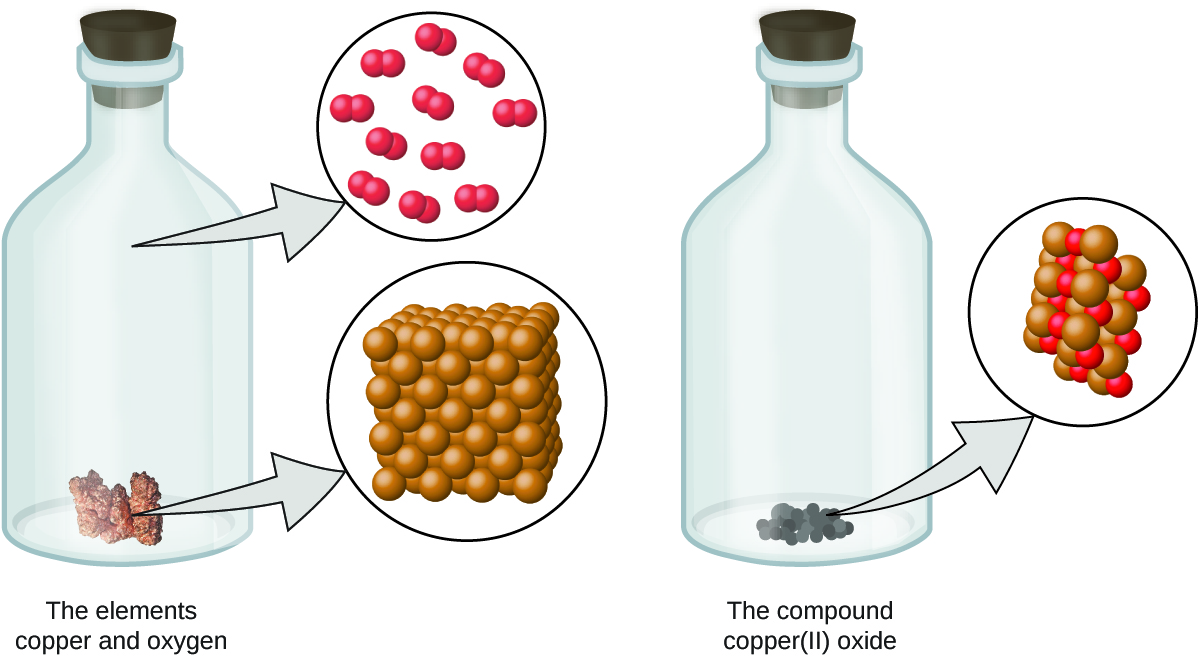
Figure 3. When the elements copper (a shiny, red-brown solid, shown here as brown spheres) and oxygen (a clear and colourless gas, shown here as red spheres) react, their atoms rearrange to form a compound containing copper and oxygen (a powdery, black solid). (Credit copper: modification of work by Chemical Elements via OpenStax) CC BY 4.0
Dalton’s atomic theory provides a microscopic explanation of the many macroscopic properties of matter that you’ve learned about. For example, if an element such as copper consists of only one kind of atom, then it cannot be broken down into simpler substances, that is, into substances composed of fewer types of atoms. And if atoms are neither created nor destroyed during a chemical change, then the total mass of matter present when matter changes from one type to another will remain constant (the law of conservation of matter).
Example 1
Testing Dalton’s Atomic Theory
In the following drawing, the green spheres represent atoms of a certain element. The purple spheres represent atoms of another element. If the spheres touch, they are part of a single unit of a compound. Does the following chemical change represented by these symbols violate any of the ideas of Dalton’s atomic theory? If so, which one?
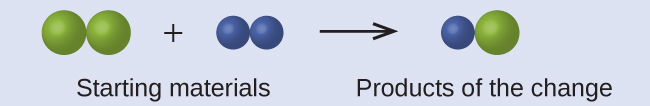
Solution
The starting materials consist of two green spheres and two purple spheres. The products consist of only one green sphere and one purple sphere. This violates Dalton’s postulate that atoms are neither created nor destroyed during a chemical change, but are merely redistributed. (In this case, atoms appear to have been destroyed.)
Check Your Learning
In the following drawing, the green spheres represent atoms of a certain element. The purple spheres represent atoms of another element. If the spheres touch, they are part of a single unit of a compound. Does the following chemical change represented by these symbols violate any of the ideas of Dalton’s atomic theory? If so, which one?
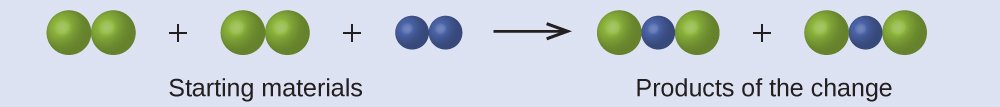
Answer:
The starting materials consist of four green spheres and two purple spheres. The products consist of four green spheres and two purple spheres. This does not violate any of Dalton’s postulates: Atoms are neither created nor destroyed, but are redistributed in small, whole-number ratios.
Dalton knew of the experiments of French chemist Joseph Proust, who demonstrated that all samples of a pure compound contain the same elements in the same proportion by mass. This statement is known as the law of definite proportions or the law of constant composition. The suggestion that the numbers of atoms of the elements in a given compound always exist in the same ratio is consistent with these observations. For example, when different samples of isooctane (a component of gasoline and one of the standards used in the octane rating system) are analyzed, they are found to have a carbon-to-hydrogen mass ratio of 5.33:1, as shown in Table 1.
| Sample | Carbon | Hydrogen | Mass Ratio |
|---|---|---|---|
| A | 14.82 g | 2.78 g | |
| B | 22.33 g | 4.19 g | |
| C | 19.40 g | 3.64 g | |
| Table 1. Constant Composition of Isooctane | |||
It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general. That is, samples that have the same mass ratio are not necessarily the same substance. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.
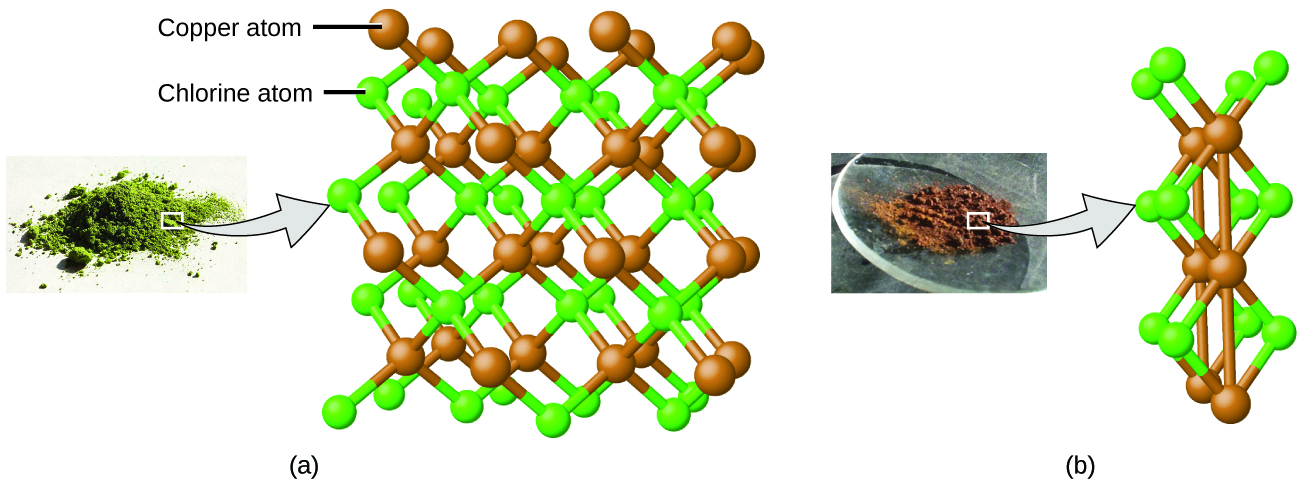
Dalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper. These ratios by themselves may not seem particularly interesting or informative; however, if we take a ratio of these ratios, we obtain a useful and possibly surprising result: a small, whole-number ratio.
This 2-to-1 ratio means that the brown compound has twice the amount of chlorine per amount of copper as the green compound.
This can be explained by atomic theory if the copper-to-chlorine ratio in the brown compound is 1 copper atom to 2 chlorine atoms, and the ratio in the green compound is 1 copper atom to 1 chlorine atom. The ratio of chlorine atoms (and thus the ratio of their masses) is therefore 2 to 1 (Figure 4).
Example 2
Laws of Definite and Multiple Proportions
A sample of compound A (a clear, colourless gas) is analyzed and found to contain 4.27 g carbon and 5.69 g oxygen. A sample of compound B (also a clear, colourless gas) is analyzed and found to contain 5.19 g carbon and 13.84 g oxygen. Are these data an example of the law of definite proportions, the law of multiple proportions, or neither? What do these data tell you about substances A and B?
Solution
In compound A, the mass ratio of carbon to oxygen is:
In compound B, the mass ratio of carbon to oxygen is:
The ratio of these ratios is:
This supports the law of multiple proportions. This means that A and B are different compounds, with A having one-half as much carbon per amount of oxygen (or twice as much oxygen per amount of carbon) as B. A possible pair of compounds that would fit this relationship would be A = CO2 and B = CO.
Check Your Learning
A sample of compound X (a clear, colourless, combustible liquid with a noticeable odour) is analyzed and found to contain 14.13 g carbon and 2.96 g hydrogen. A sample of compound Y (a clear, colourless, combustible liquid with a noticeable odour that is slightly different from X’s odour) is analyzed and found to contain 19.91 g carbon and 3.34 g hydrogen. Are these data an example of the law of definite proportions, the law of multiple proportions, or neither? What do these data tell you about substances X and Y?
Answer:
In compound X, the mass ratio of carbon to hydrogen is ![]() . In compound Y, the mass ratio of carbon to oxygen is
. In compound Y, the mass ratio of carbon to oxygen is ![]() . The ratio of these ratios is
. The ratio of these ratios is ![]() . This small, whole-number ratio supports the law of multiple proportions. This means that X and Y are different compounds.
. This small, whole-number ratio supports the law of multiple proportions. This means that X and Y are different compounds.
Key Concepts and Summary
The ancient Greeks proposed that matter consists of extremely small particles called atoms. Dalton postulated that each element has a characteristic type of atom that differs in properties from atoms of all other elements, and that atoms of different elements can combine in fixed, small, whole-number ratios to form compounds. Samples of a particular compound all have the same elemental proportions by mass. When two elements form different compounds, a given mass of one element will combine with masses of the other element in a small, whole-number ratio. During any chemical change, atoms are neither created nor destroyed.
Exercises
- In the following drawing, the green spheres represent atoms of a certain element. The purple spheres represent atoms of another element. If the spheres of different elements touch, they are part of a single unit of a compound. The following chemical change represented by these spheres may violate one of the ideas of Dalton’s atomic theory. Which one?
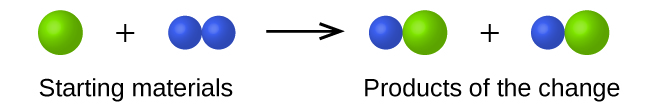
- Which postulate of Dalton’s theory is consistent with the following observation concerning the weights of reactants and products? When 100 grams of solid calcium carbonate is heated, 44 grams of carbon dioxide and 56 grams of calcium oxide are produced.
- Identify the postulate of Dalton’s theory that is violated by the following observations: 59.95% of one sample of titanium dioxide is titanium; 60.10% of a different sample of titanium dioxide is titanium.
- Samples of compound X, Y, and Z are analyzed, with results shown here.
Compound Description Mass of Carbon Mass of Hydrogen X clear, colourless liquid with strong odour 1.776 g 0.148 g Y clear, colourless liquid with strong odour 1.974 g 0.329 g Z clear, colourless liquid with strong odour 7.812 g 0.651 g Table 2. Do these data provide example(s) of the law of definite proportions, the law of multiple proportions, neither, or both? What do these data tell you about compounds X, Y, and Z?
Solutions
Answers to Odd-Numbered Exercises
1. The starting materials consist of one green sphere and two purple spheres. The products consist of two green spheres and two purple spheres. This violates Dalton’s postulate that that atoms are not created during a chemical change, but are merely redistributed.
3. This statement violates Dalton’s fourth postulate: In a given compound, the numbers of atoms of each type (and thus also the percentage) always have the same ratio.
Glossary
- atom
- smallest particle of an element that can enter into a chemical combination
- Dalton’s atomic theory
- set of postulates that established the fundamental properties of atoms
- element
- substance that is composed of a single type of atom; a substance that cannot be decomposed by a chemical change
- law of constant composition
- (also, law of definite proportions) all samples of a pure compound contain the same elements in the same proportions by mass
- law of definite proportions
- (also, law of constant composition) all samples of a pure compound contain the same elements in the same proportions by mass
- law of multiple proportions
- when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small whole numbers
Additional Activities
Video: What Is Chemistry? (2 min.)
(Free Animated Education, 2017)
Video: Sneaky Ways Chemists Are Making Our World Safer (11 min.)
(SciShow, 2021)
Media Attributions
- Figure 1: OpenStax/Rice University. (2015, March 11). Figure 2.2 [Digital image]. In Chemistry. (Section 2.1). https://openstax.org/books/chemistry/pages/2-1-early-ideas-in-atomic-theory CC BY 4.0 (Derivative of image by slgckgc [username]. Stack of pennies [Digital image]. Flickr. https://www.flickr.com/photos/131023714@N06/16401513505/.)
- Figure 2: OpenStax/Rice University. (2015, March 11). Figure 2.3 [Digital image]. In Chemistry. (Section 2.1). https://openstax.org/books/chemistry/pages/2-1-early-ideas-in-atomic-theory CC BY 4.0 (Derivative of image by Chemicalinterest [username]. Copper(II) oxide [Digital image]. Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Copper(II)_oxide.JPG. Public domain.)
- Figure 3: OpenStax/Rice University. (2015, March 11). Figure 2.4 [Digital image]. In Chemistry. (Section 2.1). https://openstax.org/books/chemistry/pages/2-1-early-ideas-in-atomic-theory CC BY 4.0 (Derivative of image by Chemical Elements.)
- Figure 4: OpenStax/Rice University. (2015, March 11). Figure 2.5 [Digital image]. In Chemistry. (Section 2.1). https://openstax.org/books/chemistry/pages/2-1-early-ideas-in-atomic-theory CC BY 4.0 (Derivative of (a) image by Benjah-bmm27 [username]. Copper(I)-chloride-sample [Digital image]. Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Copper(I)-chloride-sample.jpg. Public domain. Derivative of (b) image by Walkerma [username]. Copper(II) chloride [Digital image]. Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Iceberg_bat.jpg. Public domain.)
References
Free Animated Education. (2017, December 8). What is chemistry? [Video]. YouTube. https://youtu.be/t8x3wdXZGEY
OpenStax/Rice University. (2015, March 11). Chemistry. BC Campus. https://openstax.org/details/books/chemistry/ CC BY 4.0.
SciShow. (2021, January 17). Sneaky ways chemists are making our world safer [Video]. YouTube. https://youtu.be/yOoAwfxeEJM
Access for free at https://openstax.org/books/chemistry/pages/1-introduction

