6.2 Drawing Organic Structures
Formal Charges
Now that you have had a chance to review some basic information about atoms, orbitals, bonds, and molecules, let’s direct our attention a little more closely to the idea of charged species. You know that an ion is a molecule or atom that has an associated positive or negative charge. Copper, for example, can be found in both its neutral state (Cu0, which is the metal), or in its Cu+2 state, as a component of an ionic compound like copper carbonate (CuCO3), the green substance called “patina” that forms on the surface of copper objects.
Organic molecules can also have positive or negative charges associated with them. Consider the Lewis structure of methanol, CH3OH (methanol is the so-called “wood alcohol” that unscrupulous bootleggers sometimes sold during the prohibition days in the 1920s, often causing the people who drank it to go blind). Methanol itself is a neutral molecule, but can lose a proton to become a molecular anion (CH3O–), or gain a proton to become a molecular cation (CH3OH2+).
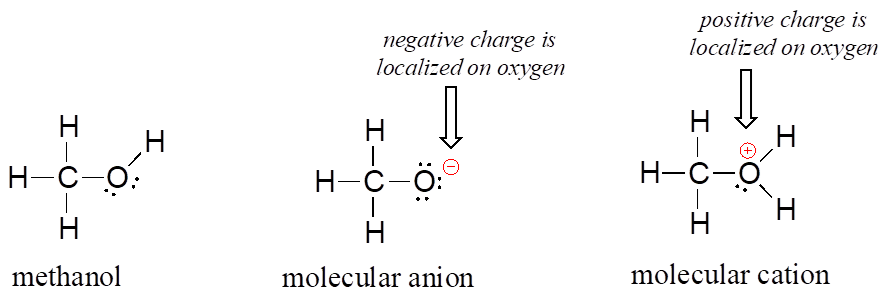
The molecular anion and cation have overall charges of -1 and +1, respectively. But we can be more specific than that—we can also state for each molecular ion that a formal charge is located specifically on the oxygen atom, rather than on the carbon or any of the hydrogen atoms.
Figuring out the formal charge on different atoms of a molecule is a straightforward process—it’s simply a matter of adding up valence electrons.
An unbound oxygen atom has 6 valence electrons. When it is bound as part of a methanol molecule, however, an oxygen atom is surrounded by 8 valence electrons: 4 nonbonding electrons (two “lone pairs”) and 2 electrons in each of its two covalent bonds (one to carbon, one to hydrogen). In the formal charge convention, we say that the oxygen “owns” all 4 nonbonding electrons. However, it only “owns” one electron from each of the two covalent bonds, because covalent bonds involve the sharing of electrons between atoms. Therefore, the oxygen atom in methanol owns 2 + 2 + (½ × 4) = 6 valence electrons.
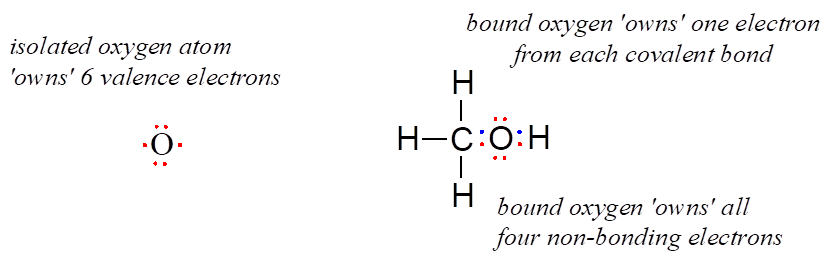
The formal charge on an atom is calculated as the number of valence electrons owned by the isolated atom minus the number of valence electrons owned by the bound atom in the molecule.
Determining the Formal Charge on an Atom in a Molecule:
formal charge =
(number of valence electrons owned by the isolated atom)
– (number of valence electrons owned by the bound atom)
or . . .
formal charge =
(number of valence electrons owned by the isolated atom)
– (number of non-bonding electrons on the bound atom)
– (½ the number of bonding electrons on the bound atom)
Using this formula for the oxygen atom of methanol, we have:
formal charge on oxygen =
(6 valence electrons on isolated atom)
– (4 non-bonding electrons)
– (½ × 4 bonding electrons)
= 6 – 4 – 2 = 0
Thus, oxygen in methanol has a formal charge of zero (in other words, it has no formal charge).
How about the carbon atom in methanol? An isolated carbon owns 4 valence electrons. The bound carbon in methanol owns (½ × 8) = 4 valence electrons:
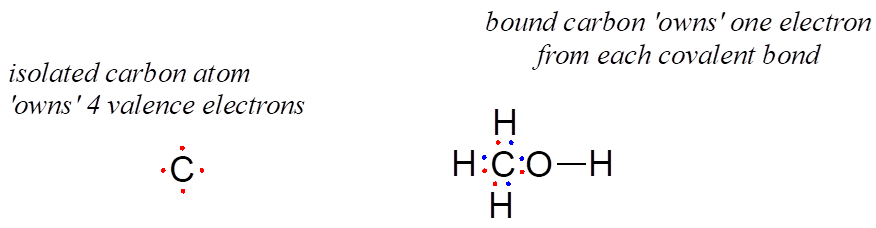
formal charge on carbon =
(4 valence electron on isolated atom)
– (0 nonbonding electrons)
– (½ × 8 bonding electrons)
= 4 – 0 – 4 = 0
. . . so the formal charge on carbon is zero.
For each of the hydrogens in methanol, we also get a formal charge of zero:
formal charge on hydrogen =
(1 valence electron on isolated atom)
– (0 nonbonding electrons)
– (½ × 2 bonding electrons)
= 1 – 0 – 1 = 0
Now, let’s look at the cationic form of methanol, CH3OH2+. The bonding picture has not changed for carbon or for any of the hydrogen atoms, so we will focus on the oxygen atom.

The oxygen owns 2 non-bonding electrons and 3 bonding elections, so the formal charge calculations becomes:
formal charge on oxygen =
(6 valence electrons in isolated atom)
– (2 non-bonding electrons)
– (½ × 6 bonding electrons)
= 6 – 2 – 3 = 1
A formal charge of +1 is located on the oxygen atom.
For methoxide, the anionic form of methanol, the calculation for the oxygen atom is:
formal charge on oxygen =
(6 valence electrons in isolated atom)
– (6 non-bonding electrons)
– (½ × 2 bonding electrons)
= 6 – 6 – 1 = -1
. . . so a formal charge of -1 is located on the oxygen atom.

A very important rule to keep in mind is that the sum of the formal charges on all atoms of a molecule must equal the net charge on the whole molecule.
When drawing the structures of organic molecules, it is very important to show all non-zero formal charges, being clear about where the charges are located. A structure that is missing non-zero formal charges is not correctly drawn, and will probably be marked as such on an exam!
At this point, thinking back to what you learned in general chemistry, you are probably asking “What about dipoles? Doesn’t an oxygen atom in an O-H bond ‘own’ more of the electron density than the hydrogen, because of its greater electronegativity?” This is absolutely correct, and we will be reviewing the concept of bond dipoles later on. For the purpose of calculating formal charges, however, bond dipoles don’t matter—we always consider the two electrons in a bond to be shared equally, even if that is not an accurate reflection of chemical reality. Formal charges are just that—a formality, a method of electron book-keeping that is tied into the Lewis system for drawing the structures of organic compounds and ions. Later, we will see how the concept of formal charge can help us to visualize how organic molecules react.
Finally, don’t be lured into thinking that just because the net charge on a structure is zero there are no atoms with formal charges: one atom could have a positive formal charge and another a negative formal charge, and the net charge would still be zero. Zwitterions, such as amino acids, have both positive and negative formal charges on different atoms:
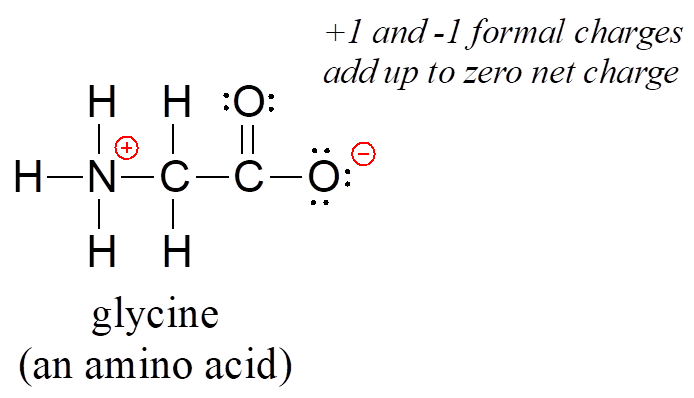
Even though the net charge on glycine is zero, it is still mandatory to show the location of the positive and negative formal charges.
Exercise 4:
Fill in all missing lone pair electrons and formal charges in the structures below. Assume that all atoms have a complete valence shell of electrons. Net charges are shown outside the brackets.
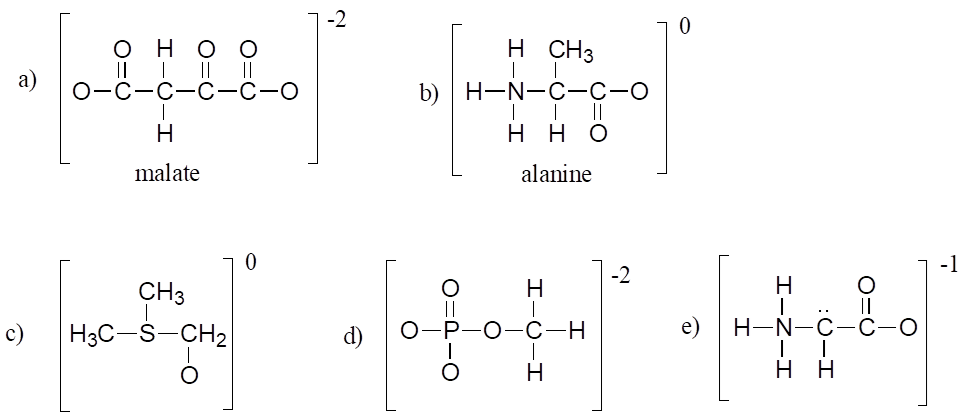
Common Bonding Patterns in Organic Structures
The electron-counting methods for drawing Lewis structures and determining formal charges on atoms are an essential starting point for a novice organic chemist, and work quite well when dealing with small, simple structures. But as you can imagine, these methods become unreasonably tedious and time-consuming when you start dealing with larger structures. It would be unrealistic, for example, to ask you to draw the Lewis structure below (of one of the four nucleoside building blocks that make up DNA) and determine all formal charges by adding up, on an atom-by-atom basis, all of the valence electrons.
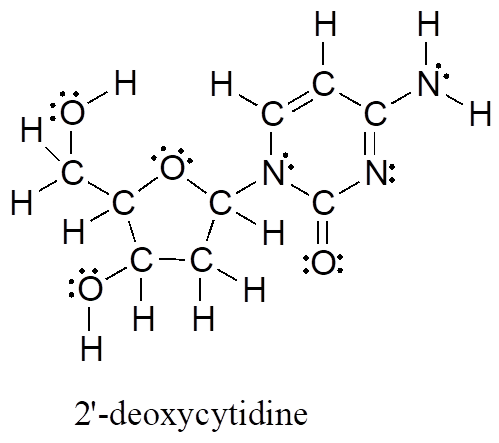
And yet, as organic chemists, and especially as organic chemists dealing with biological molecules, you will be expected soon to draw the structures of large molecules on a regular basis. Clearly, you need to develop the ability to quickly and efficiently draw large structures and determine formal charges. Fortunately, this ability is not terribly hard to come by—all it takes is learning a few shortcuts and getting some practice at recognizing common bonding patterns.
Let’s start with carbon, the most important element for organic chemists. Carbon is tetravalent, meaning that it tends to form four bonds. If you look again carefully at the structure of the DNA nucleoside 2′-deoxycytidine above, you should recognize that each carbon atom has four bonds, no lone pairs, and a formal charge of zero. Some of the carbon atoms have four single bonds, and some have one double bond and two single bonds. These are the two most common bonding patterns for carbon, along with a third option where carbon has one triple bond and one single bond.
Common Bonding Patterns for Carbon
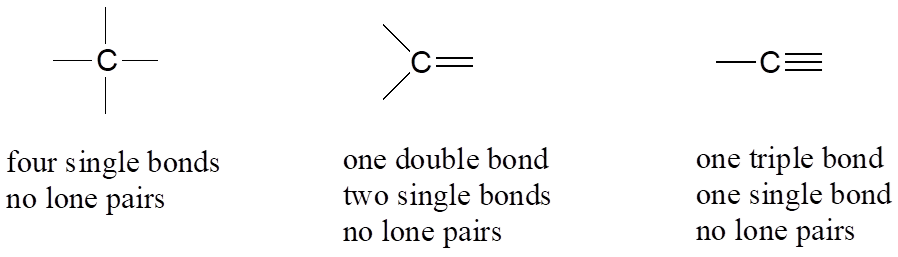
These three bonding patterns apply to most organic molecules, but there are exceptions. Here is a very important such exception: in carbon dioxide, the four bonds to the carbon atom take the form of two double bonds (O=C=O).
Carbon is also sometimes seen with a formal charge of +1 (a carbocation) or -1 (a carbanion). Notice that a carbocation does not have a full octet of valence electrons.
Additional Bonding Patterns for Carbon (High Energy Intermediate Species)
Carbocations, carbanions, and carbon radicals are very high-energy (unstable) species and thus we do not expect to see them in the structure of a stable compound. However, they are important in organic chemistry because they often form as transient (short-lived) intermediates in reactions—they form, then very quickly change into something else.
The bonding pattern for hydrogen atoms is easy: only one bond, no nonbonding electrons, and no formal charge. The exceptions to this rule are the proton (H+, just a single proton and no electrons), and the hydride ion, H–, which is a proton plus two electrons. Because we are concentrating on organic chemistry as applied to living things, however, we will not be seeing “naked” protons and hydrides as such: they are far too reactive to be present in that form in aqueous solution. Nonetheless, the idea of a proton will be very important when we discuss acid-base chemistry, and the idea of a hydride ion will become very important much later when we discuss organic oxidation and reduction reactions.
We’ll next turn to oxygen atoms. Typically, you will see an oxygen bonding in one of three ways.
Common Bonding Patterns for Oxygen:

If oxygen has two bonds and two lone pairs, as in water, it will have a formal charge of zero. If it has one bond and three lone pairs, as in hydroxide ion, it will have a formal charge of -1. If it has three bonds and one lone pair, as in hydronium ion, it will have a formal charge of +1.
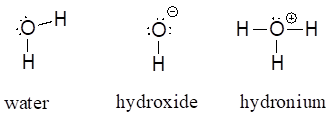
Nitrogen has two major bonding patterns: three bonds and one lone pair, or four bonds and a positive formal charge.
Common Bonding Patterns for Nitrogen:
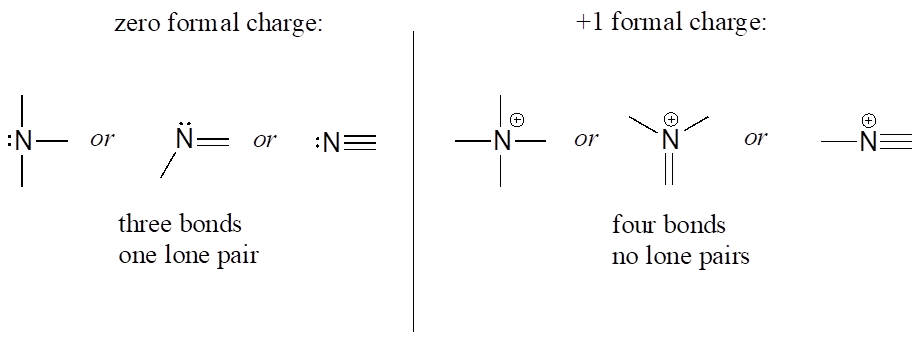
Nitrogen is occasionally also seen with two bonds, two lone pairs, and a negative formal charge—however, these species are extremely reactive and not very relevant to biological chemistry.
Two third row elements are commonly found in biological organic molecules: sulfur and phosphorus. Although both of these elements have other bonding patterns that are relevant in laboratory chemistry, in a biological context sulfur most commonly follows the same bonding/formal charge pattern as oxygen, while phosphorus is seen in a form in which it has five bonds (almost always to oxygen), no nonbonding electrons, and a formal charge of zero. Remember that atoms of elements in the third row and below in the periodic table have “expanded valence shells” with d orbitals available for bonding, and the the octet rule does not apply.
Common Bonding Pattern for Phosphorus (Phosphate)
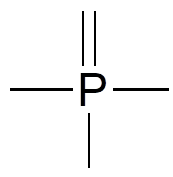
Finally, the halogens (fluorine, chlorine, bromine, and iodine) are very important in laboratory and medicinal organic chemistry, but less common in naturally occurring organic molecules. Halogens in organic chemistry usually are seen with one bond, three lone pairs, and a formal charge of zero, or as ions in solution with no bonds, four lone pairs, and a negative formal charge.
Common Bonding Pattern for Halogens:
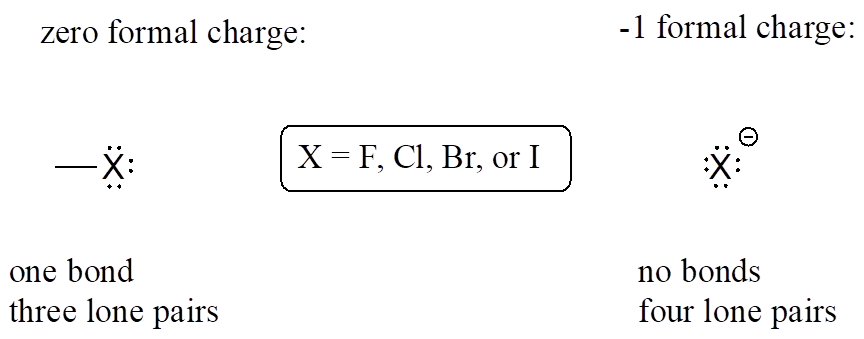
These patterns, if learned and internalized so that you don’t even need to think about them, will allow you to draw large organic structures, complete with formal charges, quite quickly.
Once you have gotten the hang of drawing Lewis structures in this way, it is not always necessary to draw lone pairs on heteroatoms, as you can assume that the proper number of electrons is present around each atom to match the indicated formal charge (or lack thereof). Often, though, lone pairs are drawn, particularly on nitrogen, if doing so helps to make an explanation more clear.
Exercise 5:
Draw one structure that corresponds to each of the following molecular formulas, using the common bonding patterns covered above. Be sure to include all lone pairs and formal charges where applicable, and assume that all atoms have a full valence shell of electrons. More than one correct answer is possible for each, so you will want to check your answers with your instructor or tutor.
- C5H10O
- C5H8O
- C6H8NO+
- C4H3O2–
Using Condensed Structures and Line Structures
If you look ahead in this and other books at the way organic compounds are drawn, you will see that the figures are somewhat different from the Lewis structures you are used to seeing in your general chemistry book. In some sources, you will see condensed structures for smaller molecules instead of full Lewis structures:
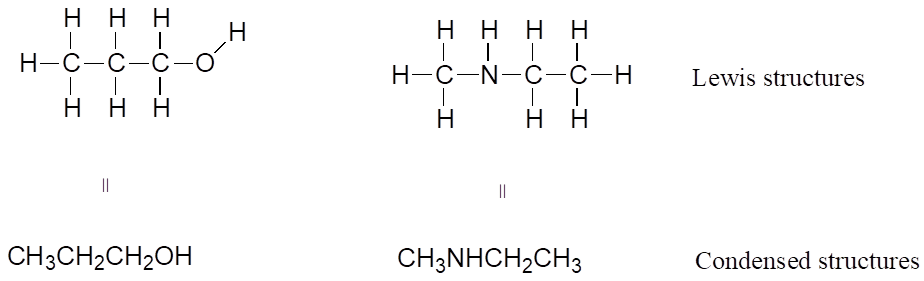
More commonly, organic and biological chemists use an abbreviated drawing convention called line structures. The convention is quite simple and makes it easier to draw molecules, but line structures do take a little bit of getting used to. Carbon atoms are depicted not by a capital C, but by a “corner” between two bonds, or a free end of a bond. Open-chain molecules are usually drawn out in a “zig-zig” shape. Hydrogens attached to carbons are generally not shown: rather, like lone pairs, they are simply implied (unless a positive formal charge is shown, all carbons are assumed to have a full octet of valence electrons). Hydrogens bonded to nitrogen, oxygen, sulfur, or anything other than carbon are shown, but are usually drawn without showing the bond. The following examples illustrate the convention.
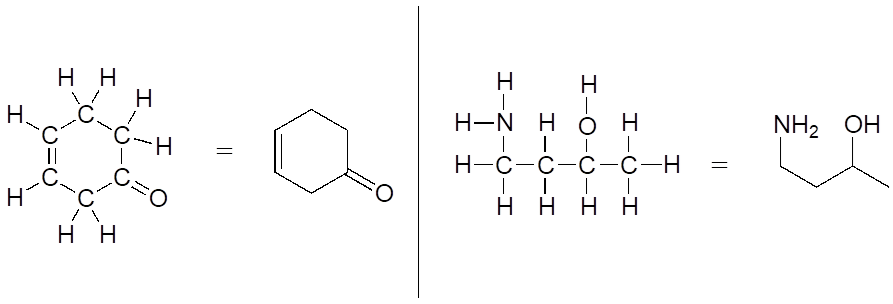
As you can see, the “pared down” line structure makes it much easier to see the basic structure of the molecule and the locations where there is something other than C-C and C-H single bonds. For larger, more complex biological molecules, it becomes impractical to use full Lewis structures. Conversely, very small molecules such as ethane should be drawn with their full Lewis or condensed structures.
Sometimes, one or more carbon atoms in a line structure will be depicted with a capital C, if doing so makes an explanation easier to follow. If you label a carbon with a C, you also must draw in the hydrogens for that carbon.
Exercise 6:
A good way to test your understanding of the line structure convention is to determine the number of hydrogen atoms in a molecule from its line structure. Do this for the structures below.
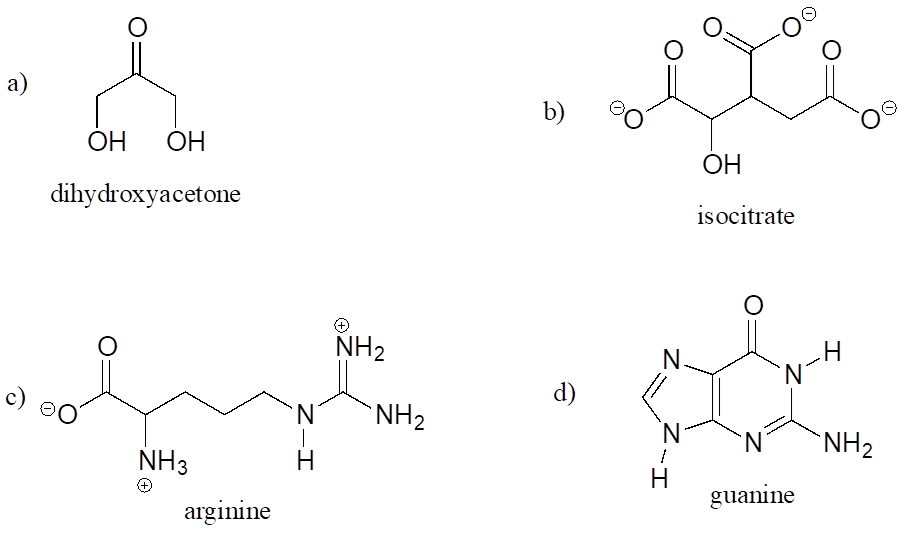
Exercise 7:
- Draw a line structure for the DNA base 2-deoxycytidine (the full structure was shown earlier).
- Draw line structures for histidine (an amino acid) and pyridoxine (Vitamin B6).
Exercise 8:
Add non-zero formal charges to the structural drawing below:
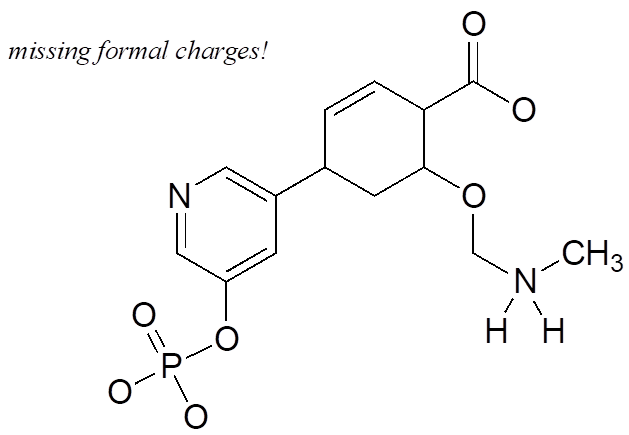
Exercise 9:
Find one example of each of the common bonding patterns specified below. Check your answers with your instructor or tutor.
- Carbon with one double bond, two single bonds, no lone pairs, and zero formal charge
- Oxygen with two single bonds, two lone pairs, and zero formal charge
- Oxygen with one double bond, two lone pairs, and zero formal charge
- Nitrogen with one double bond, two single bonds, and a +1 formal charge
- Oxygen with one single bond, three lone pairs, and a negative formal charge
Constitutional Isomers
Imagine that you were asked to draw a structure for a compound with the molecular formula C4H10. This would not be difficult—you could simply draw:
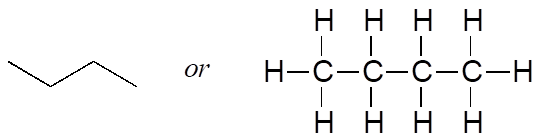
But when you compared your answer with that of a classmate, she may have drawn this structure:
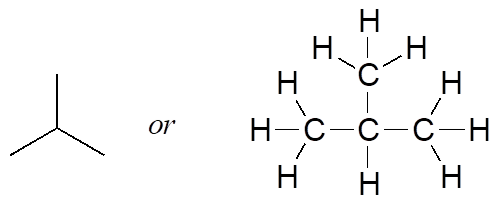
Who is correct? The answer, of course, is that both of you are. A molecular formula only tells you how many atoms of each element are present in the compound, not what the actual atom-to-atom connectivity is. There are often many different possible structures for one molecular formula. Compounds that have the same molecular formula but different connectivity are called constitutional isomers (sometimes the term “structural isomer” is also used). The Greek term “iso” means “same.”
Fructose and glucose are constitutional isomers with the molecular formula C6H12O6.
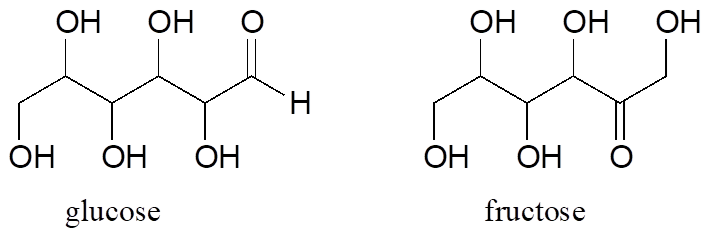
Exercise 10:
Draw a constitutional isomer of ethanol, CH3CH2OH.
Exercise 11:
Draw all of the possible constitutional isomers with the given molecular formula.
- C5H12
- C4H10O
- C3H9N
Functional Groups in Organic Compounds
Functional groups are structural units within organic compounds that are defined by specific bonding arrangements between specific atoms. The structure of capsaicin, the compound discussed in the beginning of this topic, incorporates several functional groups, labelled in the figure below and explained throughout this section.
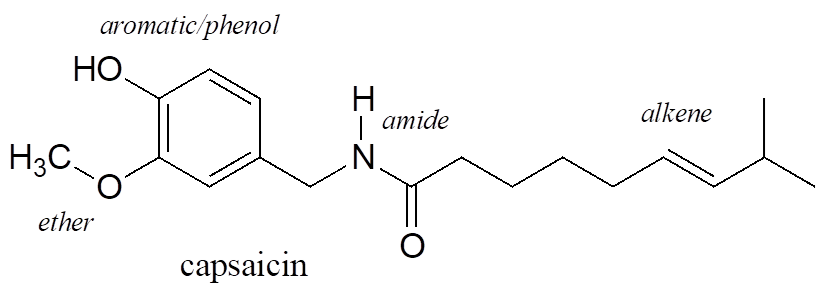
As we progress in our study of organic chemistry, it will become extremely important to be able to quickly recognize the most common functional groups, because they are the key structural elements that define how organic molecules react. For now, we will only worry about drawing and recognizing each functional group, as depicted by Lewis and line structures. Much of the remainder of your study of organic chemistry will be taken up with learning about how the different functional groups behave in organic reactions.
The “default” in organic chemistry (essentially, the lack of any functional groups) is given the term alkane, characterized by single bonds between carbon and carbon, or between carbon and hydrogen. Methane, CH4, is the natural gas you may burn in your furnace. Octane, C8H18, is a component of gasoline.
Alkanes
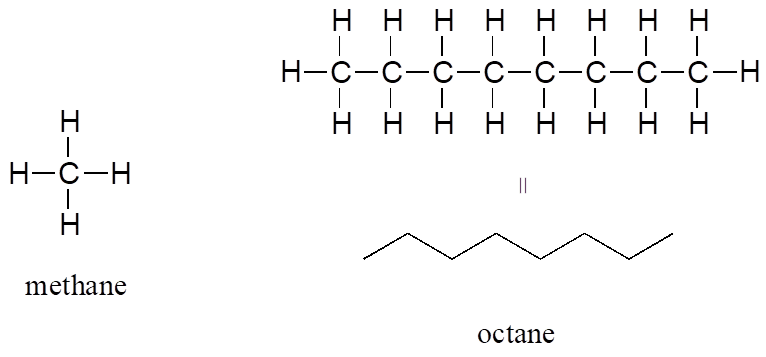
Alkenes (sometimes called olefins) have carbon-carbon double bonds, and alkynes have carbon-carbon triple bonds. Ethene, the simplest alkene example, is a gas that serves as a cellular signal in fruits to stimulate ripening. (If you want bananas to ripen quickly, put them in a paper bag along with an apple—the apple emits ethene gas, setting off the ripening process in the bananas.) Ethyne, commonly called acetylene, is used as a fuel in welding blow torches.
Alkenes and Alkynes

The bonding in alkenes is trigonal planar and in alkynes is linear. Furthermore, many alkenes can take two geometric forms: cis or trans. The cis and trans forms of a given alkene are different molecules with different physical properties because there is a very high energy barrier to rotation about a double bond. In the example below, the difference between cis and trans alkenes is readily apparent.

We will have more to say about the subject of cis and trans alkenes later.
Alkanes, alkenes, and alkynes are all classified as hydrocarbons, because they are composed solely of carbon and hydrogen atoms. Alkanes are said to be saturated hydrocarbons, because the carbons are bonded to the maximum possible number of hydrogens—in other words, they are saturated with hydrogen atoms. The double and triple-bonded carbons in alkenes and alkynes have fewer hydrogen atoms bonded to them—they are thus referred to as unsaturated hydrocarbons. Hydrogen can be added to double and triple bonds, in a type of reaction called “hydrogenation.”
The aromatic group is exemplified by benzene (which used to be a commonly used solvent on the organic lab, but which was shown to be carcinogenic), and naphthalene, a compound with a distinctive “mothball” smell. Aromatic groups are planar (flat) ring structures, and are widespread in nature.
Aromatics
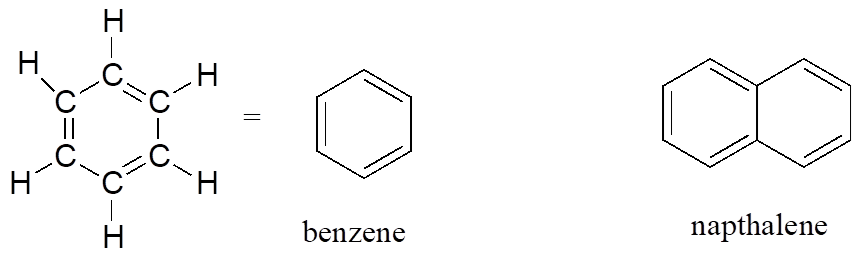
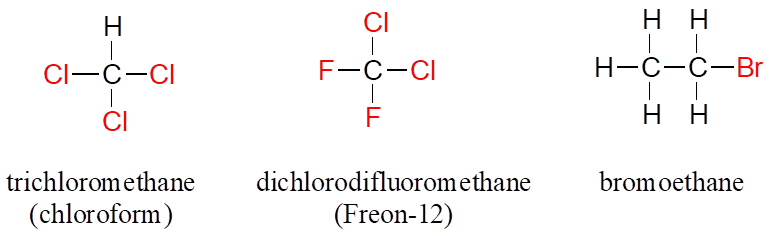
When the carbon of an alkane is bonded to one or more halogens, the group is referred to as a alkyl halide or haloalkane. Chloroform is a useful solvent in the laboratory, and was one of the earlier anesthetic drugs used in surgery. Chlorodifluoromethane was used as a refrigerant and in aerosol sprays until the late 20th century, but its use was discontinued after it was found to have harmful effects on the ozone layer. Bromoethane is a simple alkyl halide often used in organic synthesis. Alkyl halides groups are quite rare in biomolecules.
Alkyl Halides
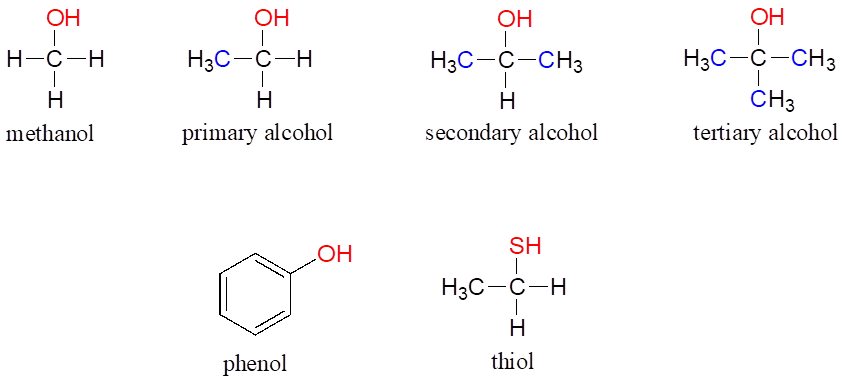
In the alcohol functional group, a carbon is single-bonded to an OH group (the OH group, by itself, is referred to as a hydroxyl). Except for methanol, all alcohols can be classified as primary, secondary, or tertiary. In a primary alcohol, the carbon bonded to the OH group is also bonded to only one other carbon. In a secondary alcohol and tertiary alcohol, the carbon is bonded to two or three other carbons, respectively. When the hydroxyl group is directly attached to an aromatic ring, the resulting group is called a phenol. The sulfur analog of an alcohol is called a thiol (from the Greek thio, for sulfur)
Alcohols, Phenols, and Thiols
Note that the definition of a phenol states that the hydroxyl oxygen must be directly attached to one of the carbons of the aromatic ring. The compound below, therefore, is not a phenol—it is a primary alcohol.
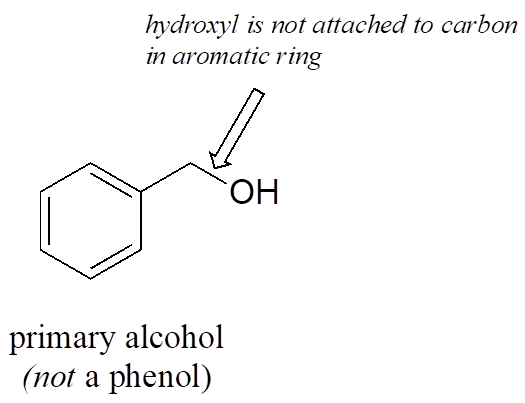
The distinction is important, because as we will see later, there is a significant difference in the reactivity of alcohols and phenols.
The deprotonated forms of alcohols, phenols, and thiols are called alkoxides, phenolates, and thiolates, respectively. A protonated alcohol is an oxonium ion.

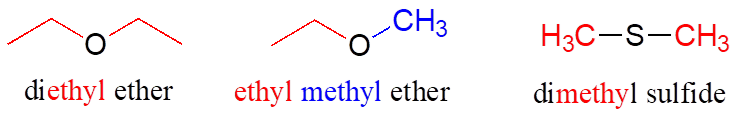
In an ether functional group, a central oxygen is bonded to two carbons. Below is the structure of diethyl ether, a common laboratory solvent and also one of the first compounds to be used as an anesthetic during operations. The sulfur analog of an ether is called a thioether or sulfide.
Ethers and Sulfides
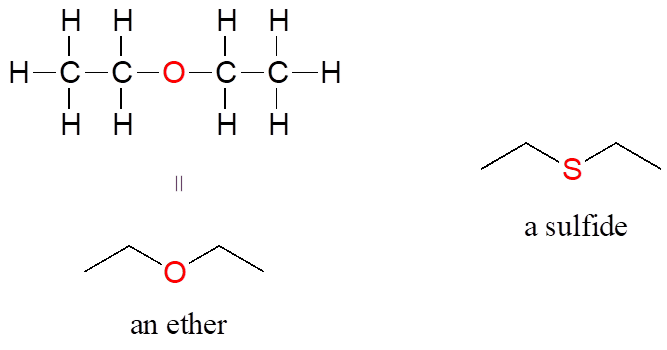
Amines are characterized by nitrogen atoms with single bonds to hydrogen and carbon. Just as there are primary, secondary, and tertiary alcohols, there are primary, secondary, and tertiary amines. Ammonia is a special case with no carbon atoms.
One of the most important properties of amines is that they are basic, and are readily protonated to form ammonium cations. In the case where a nitrogen has four bonds to carbon (which is somewhat unusual in biomolecules), it is called a quaternary ammonium ion.
Amines
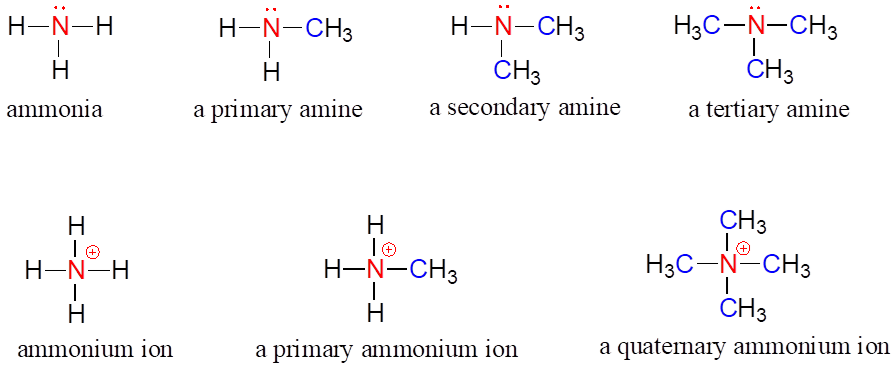
Note: Do not be confused by how the terms “primary,” “secondary,” and “tertiary” are applied to alcohols and amines—the definitions are different. In alcohols, what matters is how many other carbons the alcohol carbon is bonded to, while in amines, what matters is how many carbons the nitrogen is bonded to.
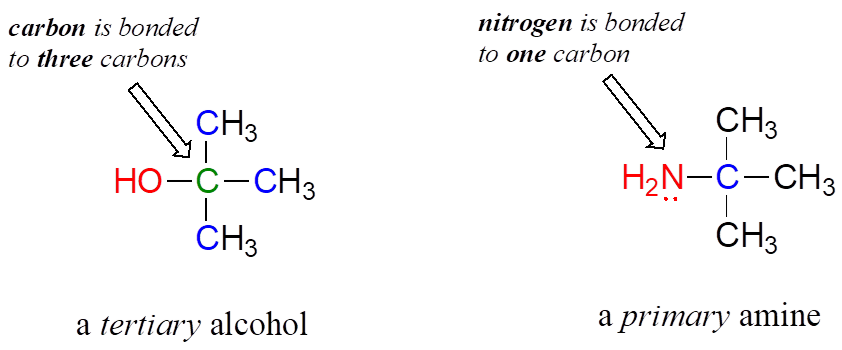
Phosphate and its derivative functional groups are ubiquitous in biomolecules. Phosphate linked to a single organic group is called a phosphate ester; when it has two links to organic groups it is called a phosphate diester. A linkage between two phosphates creates a phosphate anhydride.
Phosphate Functional Groups
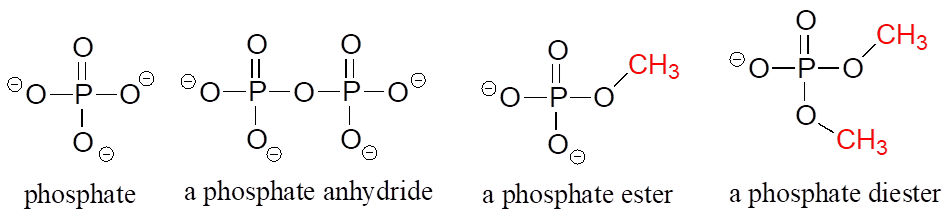
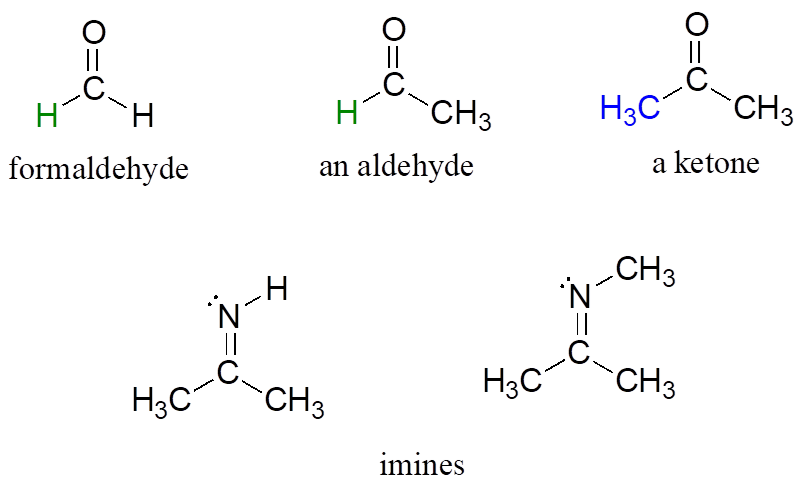
There is a number of functional groups that contain a carbon-oxygen double bond, which is commonly referred to as a carbonyl. Ketones and aldehydes are two closely related carbonyl-based functional groups that react in very similar ways. In a ketone, the carbon atom of a carbonyl is bonded to two other carbons. In an aldehyde, the carbonyl carbon is bonded on one side to a hydrogen, and on the other side to a carbon. The exception to this definition is formaldehyde, in which the carbonyl carbon has bonds to two hydrogens.
A group with a carbon-nitrogen double bond is called an imine, or sometimes a Schiff base (we will use the term “imine”).
Aldehydes, Ketones, and Imines
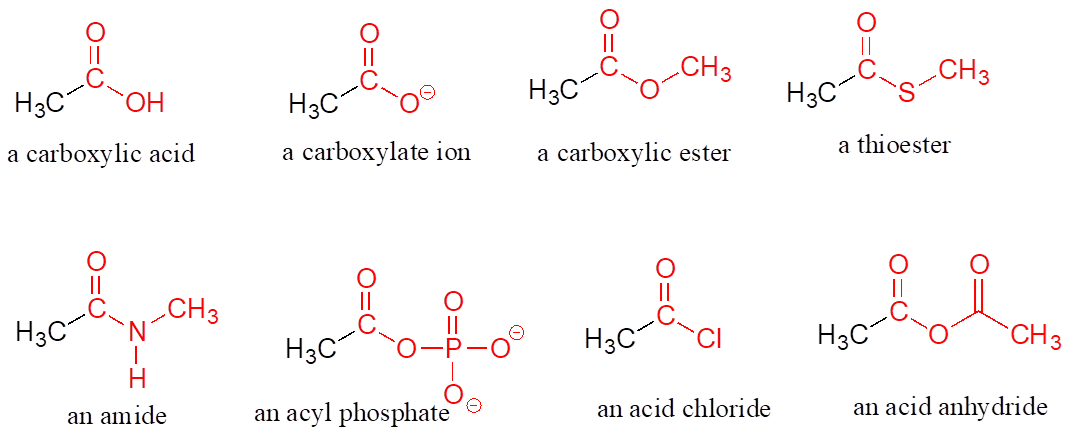
When a carbonyl carbon is bonded on one side to a carbon (or hydrogen) and on the other side to an oxygen, nitrogen, or sulfur, the functional group is considered to be one of the “carboxylic acid derivatives,” a designation that describes a set of related functional groups. The eponymous member of this family is the carboxylic acid functional group, in which the carbonyl is bonded to a hydroxyl group. The conjugate base of a carboxylic acid is a carboxylate. Other derivatives are carboxylic esters (usually just called “esters”), thioesters, amides, acyl phosphates, acid chlorides, and acid anhydrides. With the exception of acid chlorides and acid anhydrides, the carboxylic acid derivatives are very common in biological molecules and/or metabolic pathways.
Carboxylic Acid Derivatives
Finally, a nitrile group is characterized by a carbon triple-bonded to a nitrogen.
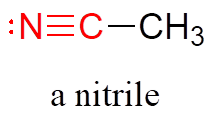
A single compound often contains several functional groups, particularly in biological organic chemistry. The six-carbon sugar molecules glucose and fructose, for example, contain aldehyde and ketone groups, respectively, and both contain five alcohol groups (a compound with several alcohol groups is often referred to as a “polyol”).
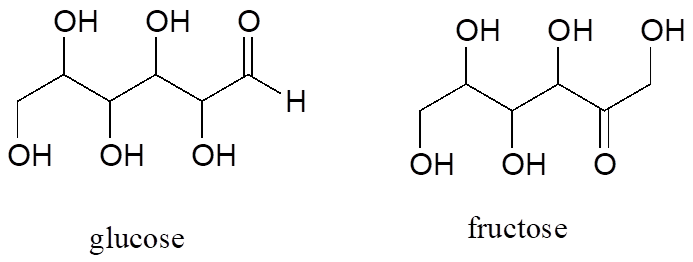
The hormone testosterone, the amino acid phenylalanine, and the glycolysis metabolite dihydroxyacetone phosphate all contain multiple functional groups, as labelled below.

While not in any way a complete list, this section has covered most of the important functional groups that we will encounter in biological organic chemistry.
Exercise 12:
Identify the functional groups (other than alkanes) in the following organic compounds. State whether alcohols and amines are primary, secondary, or tertiary.

Exercise 13:
Draw one example each of compounds fitting the descriptions below, using line structures. Be sure to designate the location of all non-zero formal charges. All atoms should have complete octets (phosphorus may exceed the octet rule). There are many possible correct answers for these, so be sure to check your structures with your instructor or tutor.
- A compound with molecular formula C6H11NO that includes alkene, secondary amine, and primary alcohol functional groups
- An ion with molecular formula C3H5O6P 2- that includes aldehyde, secondary alcohol, and phosphate functional groups.
- A compound with molecular formula C6H9NO that has an amide functional group, and does not have an alkene group
Naming Organic Compounds
A system has been devised by the International Union of Pure and Applied Chemistry (IUPAC, usually pronounced eye-you-pack) for naming organic compounds. While the IUPAC system is convenient for naming relatively small, simple organic compounds, it is not generally used in the naming of biomolecules, which tend to be quite large and complex. It is, however, a good idea (even for biologists) to become familiar with the basic structure of the IUPAC system, and be able to draw simple structures based on their IUPAC names.
Naming an organic compound usually begins with identifying what is referred to as the “parent chain,” which is the longest straight chain of carbon atoms. We’ll start with the simplest straight-chain alkane structures. CH4 is called methane, and C2H6 ethane. The table below continues with the names of longer straight-chain alkanes: be sure to commit these to memory, as they are the basis for the rest of the IUPAC nomenclature system (and are widely used in naming biomolecules as well).
Names for straight-chain alkanes:
|
Number of carbons |
Name
|
|
1 |
methane |
|
2 |
ethane |
|
3 |
propane |
|
4 |
butane |
|
5 |
pentane |
|
6 |
hexane |
|
7 |
heptane |
|
8 |
octane |
|
9 |
nonane |
|
10 |
decane |
Substituents branching from the main parent chain are located by a carbon number, with the lowest possible numbers being used (for example, notice in the example below that the compound on the left is named 1-chlorobutane, not 4-chlorobutane). When the substituents are small alkyl groups, the terms methyl, ethyl, and propyl are used.
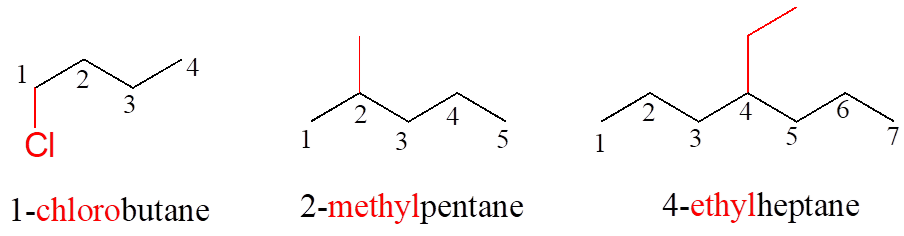
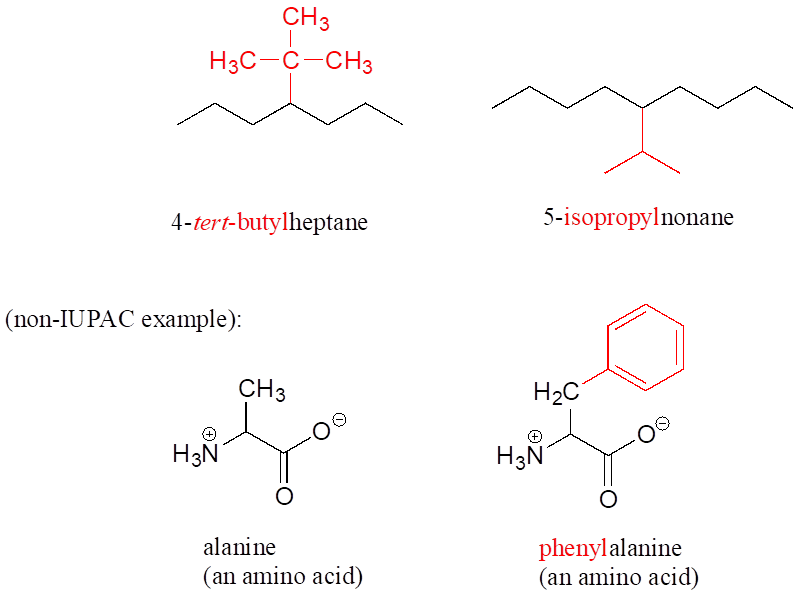
Other common names for hydrocarbon substituent groups isopropyl, tert-butyl, and phenyl.
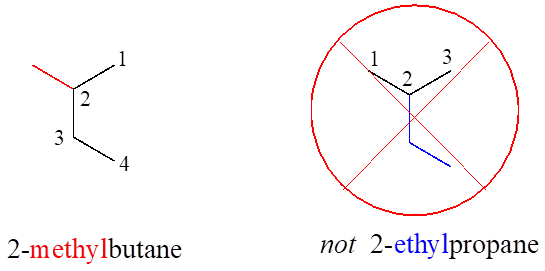
Notice in the example below, an “ethyl group” (in blue) is not treated as a substituent, rather it is included as part of the parent chain, and the methyl group is treated as a substituent. The IUPAC name for straight-chain hydrocarbons is always based on the longest possible parent chain, which in this case is four carbons, not three.
Cyclic alkanes are called cyclopropane, cyclobutane, cyclopentane, cyclohexane, and so on.
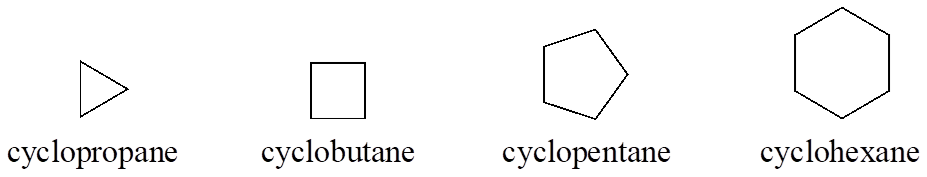
In the case of multiple substituents, the prefixes di, tri, and tetra are used.
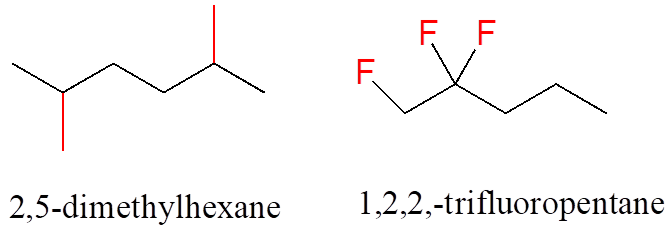
Functional groups have characteristic suffixes. Alcohols, for example, have “ol” appended to the parent chain name, along with a number designating the location of the hydroxyl group. Ketones are designated by “one.”
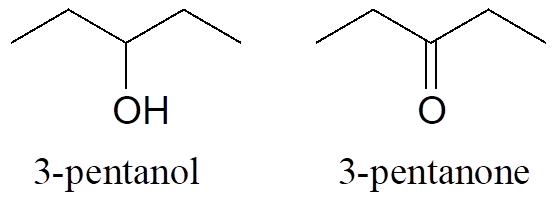
Alkenes are designated with an “ene” ending, and when necessary the location and geometry of the double bond are indicated. Compounds with multiple double bonds are called dienes, trienes, etc.
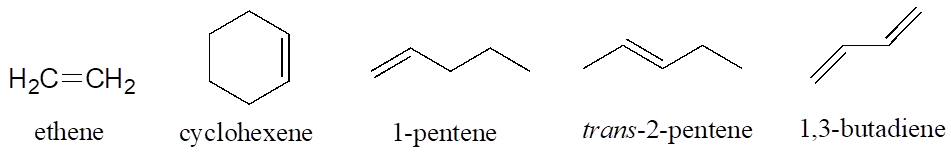
Some groups can only be present on a terminal carbon, and thus a locating number is not necessary: aldehydes end in “al,” carboxylic acids in “oic acid,” and carboxylates in “oate.”

Ethers and sulfides are designated by naming the two groups on either side of the oxygen or sulfur.
If an amide has an unsubstituted –NH2 group, the suffix is simply “amide.” In the case of a substituted amide, the group attached to the amide nitrogen is named first, along with the letter “N” to clarify where this group is located. Note that the structures below are both based on a three-carbon (propan) parent chain.
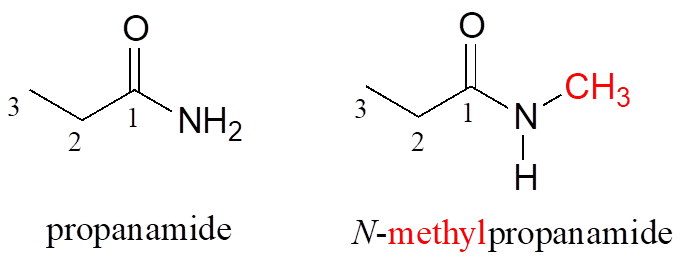
For esters, the suffix is “oate.” The group attached to the oxygen is named first.

All of the examples we have seen so far have been simple in the sense that only one functional group was present on each molecule. There are of course many more rules in the IUPAC system, and as you can imagine, the IUPAC naming of larger molecules with multiple functional groups, ring structures, and substituents can get very unwieldy very quickly. The illicit drug cocaine, for example, has the IUPAC name “methyl (1R,2R,3S,5S)-3-(benzoyloxy)-8-methyl-8-azabicyclo[3.2.1] octane-2-carboxylate” (this name includes designations for stereochemistry, which is a structural issue that we will not tackle until later).
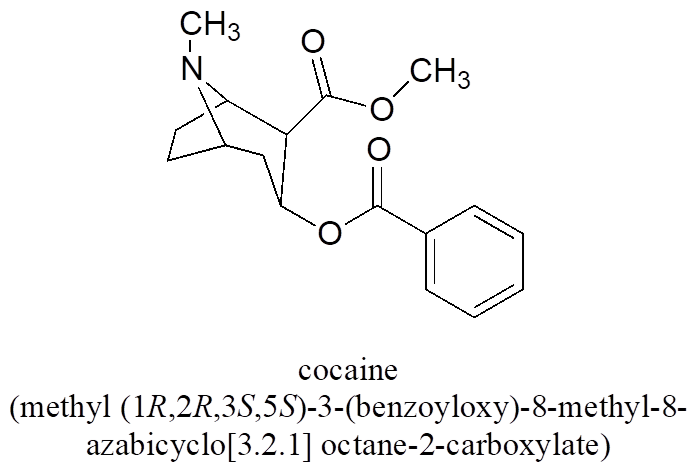
You can see why the IUPAC system is not used very much in biological organic chemistry—the molecules are just too big and complex. A further complication is that, even outside of a biological context, many simple organic molecules are known almost universally by their “common,” rather than IUPAC names. The compounds acetic acid, chloroform, and acetone are only a few examples.
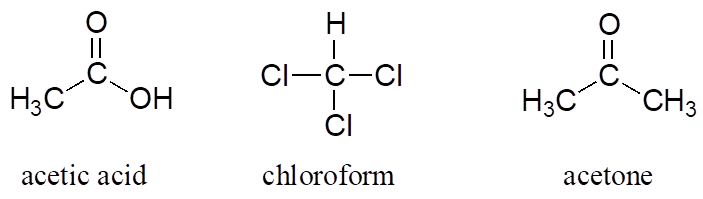
In biochemistry, nonsystematic names (like “cocaine,” “capsaicin,” “pyruvate,” or “ascorbic acid”) are usually used, and when systematic nomenclature is employed it is often specific to the class of molecule in question: different systems have evolved, for example, for fats and for carbohydrates. We will not focus very intensively in this text on IUPAC nomenclature or any other nomenclature system, but if you undertake a more advanced study in organic or biological chemistry you may be expected to learn one or more naming systems in some detail.
Exercise 14:
Give IUPAC names for acetic acid, chloroform, and acetone.
Exercise 15:
Draw line structures of the following compounds, based on what you have learned about the IUPAC nomenclature system:
- Methylcyclohexane
- 5-methyl-1-hexanol
- 2-methyl-2-butene
- 5-chloropentanal
- 2,2-dimethylcyclohexanone
- 4-penteneoic acid
- N-ethyl-N-cyclopentylbutanamide
Abbreviated Organic Structures
Often when drawing organic structures, chemists find it convenient to use the letter “R” to designate part of a molecule outside of the region of interest. If we just want to refer in general to a functional group without drawing a specific molecule, for example, we can use “R“ groups to focus attention on the group of interest:
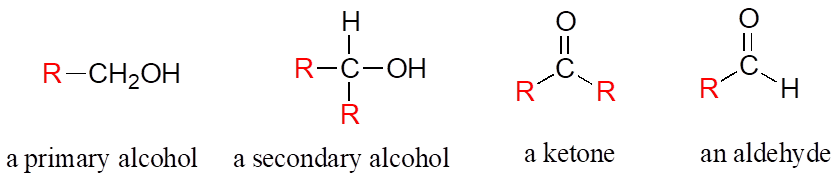
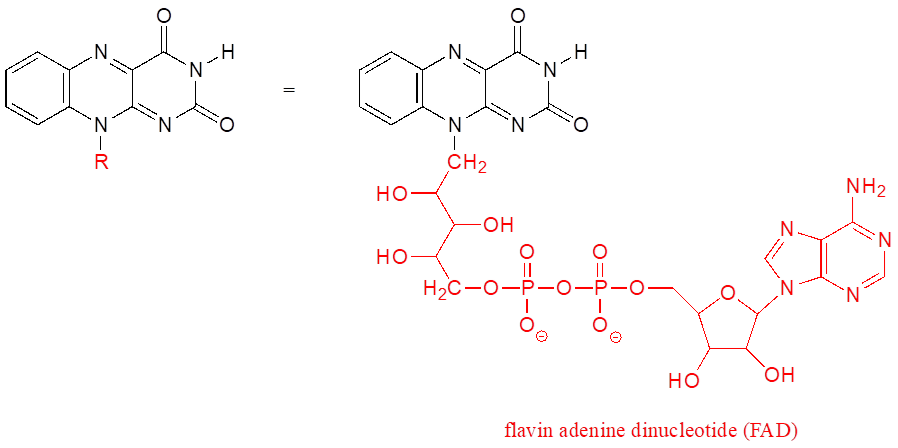
The R group is a convenient way to abbreviate the structures of large biological molecules, especially when we are interested in something that is occurring specifically at one location on the molecule. For example, looking at biochemical oxidation-reduction reactions involving the flavin molecule, we can abbreviate a large part of the flavin structure which does not change at all in the reactions of interest:
As an alternative, we can use a “break” symbol to indicate that we are looking at a small piece or section of a larger molecule. This is used commonly in the context of drawing groups on large polymers such as proteins or DNA.

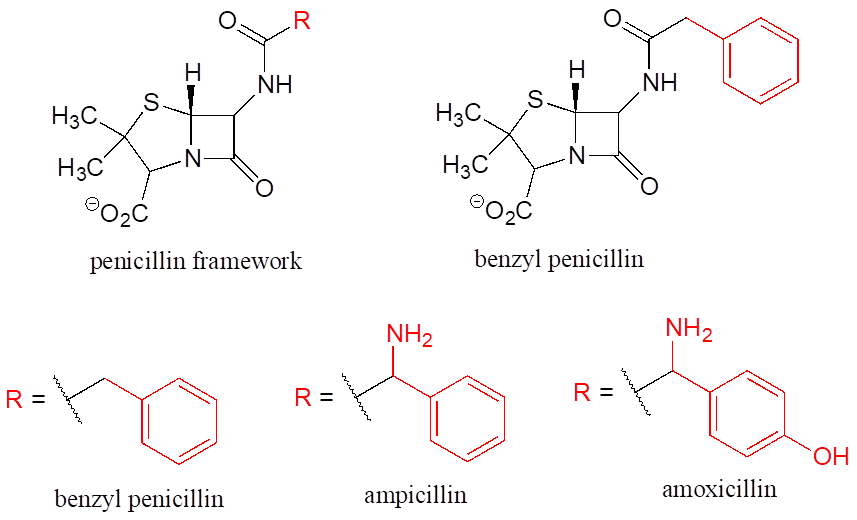
Finally, R groups can be used to concisely illustrate a series of related compounds, such as the family of penicillin-based antibiotics.
Using abbreviations appropriately is a very important skill to develop when studying organic chemistry in a biological context, because although many biomolecules are very large and complex (and take forever to draw!), usually we are focusing on just one small part of the molecule where a change is taking place. As a rule, you should never abbreviate any atom involved in a bond-breaking or bond-forming event that is being illustrated: only abbreviate that part of the molecule which is not involved in the reaction of interest. For example, carbon #2 in the reactant/product below most definitely is involved in bonding changes, and therefore should not be included in the “R” group.
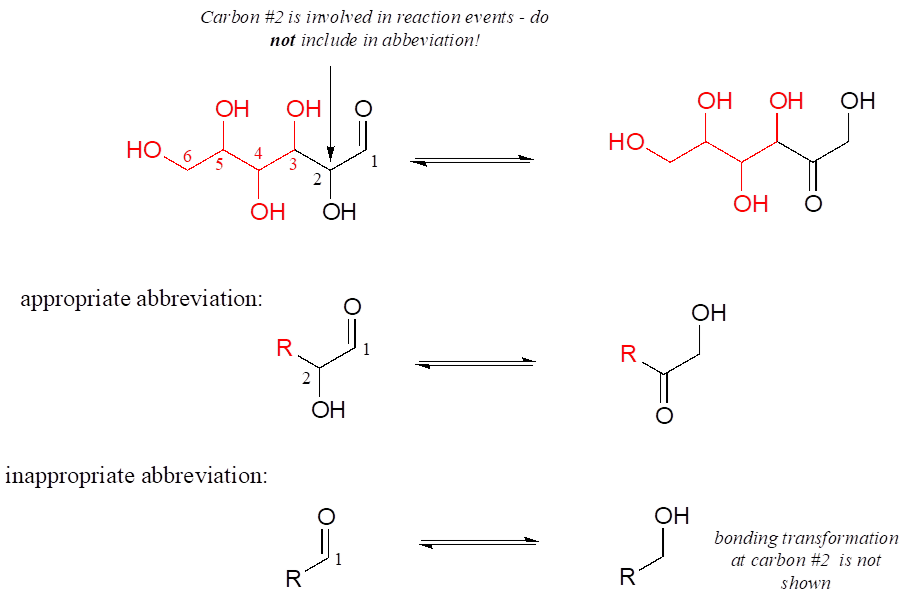
If you are unsure whether to draw out part of a structure or abbreviate it, the safest thing to do is to draw it out.
Exercise 16:
- If you intend to draw out the chemical details of a reaction in which the methyl ester functional group of cocaine (see earlier figure) was converted to a carboxylate plus methanol, what would be an appropriate abbreviation to use for the cocaine structure (assuming that you only wanted to discuss the chemistry specifically occurring at the ester group)?
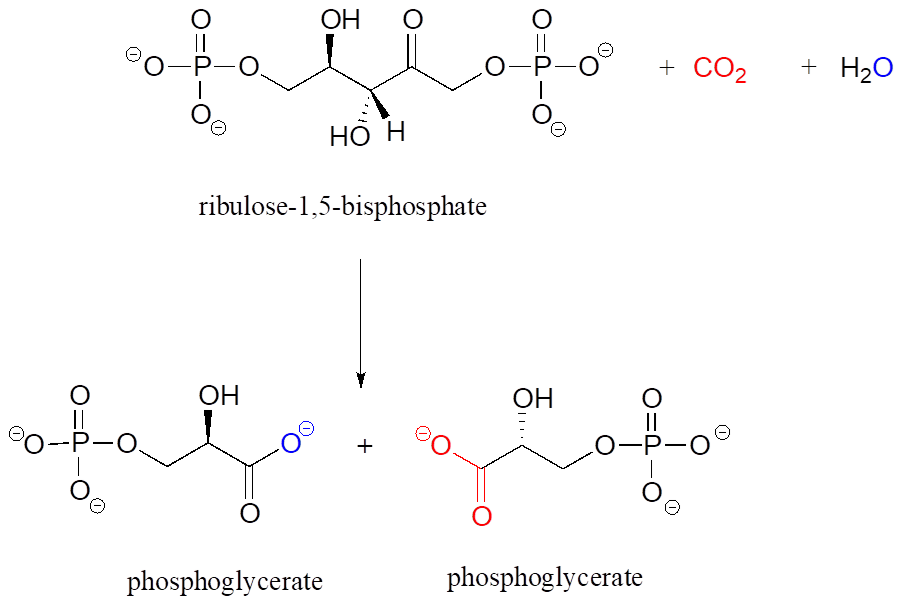
- Below is the (somewhat complicated) reaction catalyzed by an enzyme known as “rubisco,” by which plants “fix” carbon dioxide. Carbon dioxide and the oxygen of water are coloured red and blue respectively to help you see where those atoms are incorporated into the products. Propose an appropriate abbreviation for the starting compound (ribulose 1,5-bisphosphate), using two different “R” groups, R1 and R2.
Summary of Key Concepts
Before you move on to the next section, you should:
Thoroughly review the fundamental principles of atomic structure and electron configuration, and review the rules for drawing Lewis structures.
- Be very familiar with the common bonding patterns in organic molecules—you should be able to quickly recognize where lone pairs exist, even when they are not drawn explicitly, and you should be able to readily recognize incorrectly drawn structures—for example, when carbon is drawn with five bonds.
- Be able to determine the formal charge on all atoms of a compound—with practice, you should be able to look at an organic structure and very rapidly recognize when there is a formal charge on a carbon, oxygen, or nitrogen.
- Become adept at interpreting and drawing line structures for organic molecules (line structures will be used almost exclusively for the remainder of this textbook). A good test is to determine the molecular formula of a molecule from a line structure.
- Understand the meaning of constitutional isomer, and be able to recognize and/or draw constitutional isomers of a given compound.
- Be able to recognize and come up with your own examples of the most important functional groups in organic chemistry.
- Be familiar with the basic rules of the IUPAC nomenclature system, at the level presented in this textbook, and be able to draw a structure based on its IUPAC name.
- Understand how and when to use abbreviated organic structures appropriately.
- You need not memorize in detail the structures of common classes of biological molecules illustrated (fats, isoprenoids, carbohydrates, proteins, and nucleic acids), but you should be able to recognize examples when you see them.
- As always, you should be familiar with the meaning of all of the terms written in bold.
Additional Activities
Watch the videos below which go through the rules for naming and drawing organic molecules
Video: Naming Organic Compounds Part 1 (14 min. 37 sec.)
(Leah4sci, 2011a)
Video: Naming Organic Compounds Part 2 (14 min. 30 sec.)
(Leah4sci, 2011b)
Video: Organic Chemistry Drawing Structures – Bond Line, Skeletal, and Condensed Structural Formulas (31 min.)
(The Organic Chemistry Tutor, 2016)
Optional activity: For a free website you can use to draw organic molecules online, please visit PubChem Sketcher or MolView. This can help you to practise using software such as ChemDoodle and ChemDraw in the future!
Optional activity: Please visit openmolecules.org’s “Name To Structure” where you are able to easily convert IUPAC names, common names, SMILES codes, CAS numbers, and other identifiers into chemical structures—you can practise your naming and drawing of organic molecules easily with this online tool.
Watch the following video which discusses the naming of isomers of organic molecules
Video: Drawing Isomers of Organic Molecules (8 min. 21 sec.)
(Pennington, 2015)
Optional activity: For a fun and educational gaming experience, you can download the App “Chemical Substances: Organic and Inorganic Chemistry by Andrew Solovyev” to practise naming and identifying isomers of organic compounds.
Optional activity: Please visit ChemSpider’s “Structure Search” for an interactive site where you can draw structures and generate a display of their isomers where applicable.
Reference
UMM Digital Well. (2019, July). Organic chemistry with a biological emphasis volume I. University of Minnesota. https://digitalcommons.morris.umn.edu/chem_facpubs/1/ CC BY-NC-SA 4.0.


